whitepaper:
Unveiling the biotech vendor selection process

Get the scoop on ranking and selecting vendors
If you haven’t yet downloaded our whitepaper – The biotech blueprint for ranking and selecting vendors, this blog written by our Joao Incio, Bio Sector Lead is sure to whet your appetite by helping you to understand buyer behaviors and learn about the CDMO/CRO decision-making process and key criteria typically used to inform vendor selection.
The biotech buyer journey
Biotech companies heavily rely on outsourcing for development and manufacturing activities.
ramarketing recently explored the process typically used by biotech companies to select Contract Development and Manufacturing Organizations (CDMOs), Contract Manufacturing Organizations (CMOs), and Contract Research Organizations (CROs).
Following the analysis of a sample of biotech senior management employee interviews, ramarketing summarized the step-by-step approach typically used in the CDMO/CMO/CRO selection process and identified the predominant factors considered when selecting a vendor.
Download the full whitepaper here
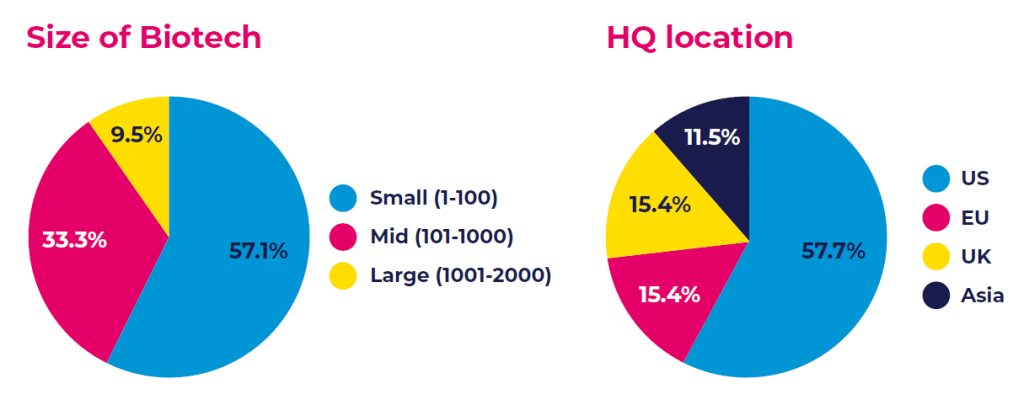
Most of the analyzed companies were small and mid-size biotechs, with a roughly equal share of public and private companies based predominantly in the US and Europe.
Based on interviews conducted with representatives from these companies, mostly C-suite Executives, VPs, and Senior Directors, predominantly in manufacturing and clinical operations, the whitepaper describes the buyer’s journey.
Step-by-step vendor selection
This analysis revealed that although there may be some variability in how biotechs conduct CDMO/CRO vendor selection, it typically follows a stepped approach.
The process is initiated with pre-selection, and internal planning – where a cross-functional technical team is formed, and a matrix/scoring system (using spreadsheets or software) including parameters for decision-making is prepared.
Subsequently, biotechs identify vendors mostly via awareness/prior experience and leveraging their networks, producing a shortlist of 4-5 to whom an RFP is sent, requesting critical information for decision-making.
Meetings usually include 2-3 vendors, and a decision is made following a review of the final proposals.
Reputation is everything
“Our area is small so when we’re networking and we hear ‘This team is really good,’ we listen. If you get a bad name, that’s it — you’re finished.”
CEO, Small Biotech
Decision makers vary according to company size and vendor sought.
For CDMO/CMOs, the person driving the process includes senior CMC employees, from Associate Director all the way to Senior VP/Executive Director, with input from internal SMEs and colleagues in the CMC, regulatory, quality, and supply chain departments.
In the case of CROs, the Head/VP of Clinical Operations will drive the selection process, leveraging input from clinical research consultants, Chief Medical Officer (CMO), Medical/Scientific Affairs, CMC, and Regulatory/Safety. The CEO, CMO, and Clin Ops Head/VP are the key decision makers.
Tangible and intangible criteria inform the selection
Criteria used by biotechs for selection were split between tangible criteria (such as expertise, experience, capabilities, and capacity) and intangibles (like accessibility and responsiveness, shared cultural values, attitude and dedication, honesty, and transparency).
Expertise, capabilities, and experience ranked the highest, with cost coming in third.
A second tier of criteria cited by interviewees included reputation/credibility, quality, and production capacity/timelines.
Vendor selection is a dating game
“We look at a mix of hard and soft competencies. We are looking for a partner to have a successful relationship with; this is unspoken. We need to hit it off.”
Senior Director, Mid-sized Biotech
An understanding of what drives biotech companies to select specific outsourcing partners will be critical to ensure that CDMOs and CROs position themselves to be attractive partners and better serve biotechnology companies.
Download the full whitepaper here to discover more about the ranking and selection process.
For analysis tailored to a specific CDMO/CRO customer segment, please contact our commercial team to discuss your research and strategy needs.
+44 (0) 191 222 1242
commercial@ramarketingpr.com
Get regular updates in your inbox
Fields marked with a * are required